Reading time: 10min.
Borer Chemie, 29. September 2023
Phosphate-free cleaners for the reprocessing of medical devices
So why are phosphates so popular?
With their multiple functions, phosphates are extremely popular as ingredients in water-based detergents and cleaning products. On the one hand, they ensure that the hardness components contained in the water are complexed - i.e. bound - while at the same time guaranteeing the ideal pH value for cleaning. On the other hand, they suspend dirt particles and thus prevent them from adhering to the surfaces to be cleaned. These properties are crucial for the strong cleaning performance of products containing phosphates.
And the other side of the coin?
Worldwide, 8.8 megatons of phosphorus enter the world's oceans via rivers every year. And models predict a further increase in this leaching by 2051. Excessive phosphates in water can have serious effects on the ecosystem. Phosphorus is an essential mineral for the growth of plants and algae. However, if excessive quantities of it enter bodies of water, this leads to surplus algae growth.
 Algae formation due to too-high phosphate concentration in the water
Algae formation due to too-high phosphate concentration in the water
In the worst-case scenario, this excessive growth can lead to the open water areas and bottom zones of the water body no longer being sufficiently supplied with light, as they are covered by a dense blanket of algae. The result is an oxygen deficiency, as less oxygen is produced by the reduced photosynthesis of the aquatic plants, but the same amount of oxygen is still consumed by the aquatic organisms. This leads to an ever-depleting level of oxygen. This effect, known as eutrophication, is popularly known as the "biological death" of a body of water and usually results in a mass die-off of plants and animals.
Since 1960, the number of dead zones where the sea bed is oxygen-deficient has doubled every decade (1960: 10 areas; 2008: 405 areas)2). The three largest dead zones are in the Baltic Sea (up to 84,000 km²), the Black Sea (up to 40,000 km²) and the Gulf of Mexico (up to 22,000 km²). In the long term, this eutrophication will reduce our planet's biodiversity and have a noticeable impact on our lives.
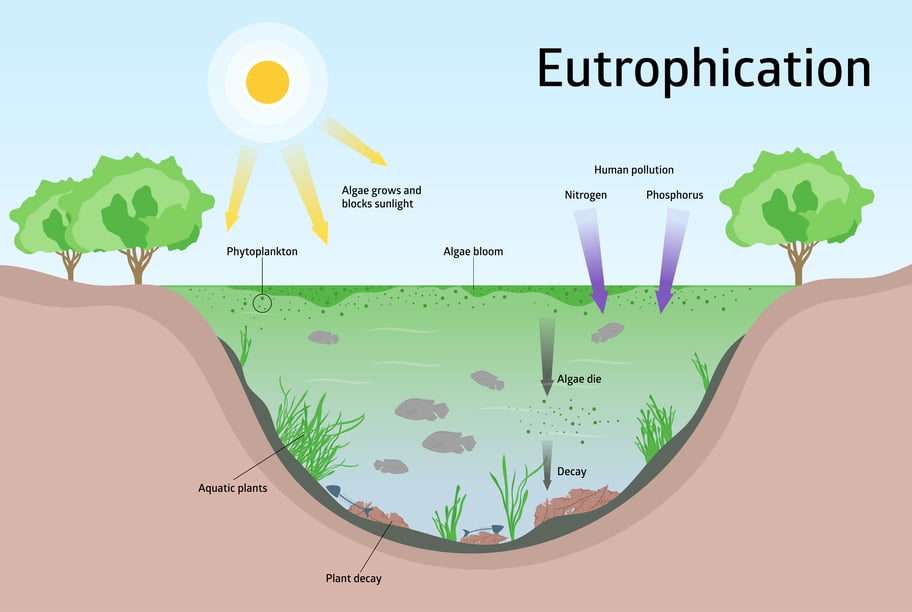
Process of eutrophication in water bodies
Phosphate-free - for the sake of the environment
The search for substitutes that have the same effect as phosphates in cleaning agents is a challenge. It takes a whole assortment of building blocks to achieve the desired result.
Our product developers have been working intensively on this topic for many years. And the news is good: almost our entire product range for the reprocessing of medical devices is now phosphate-free. From the effective pre-cleaning foam deconex® FOAM ACTIVE and FOAM PLUS to the manual cleaner deconex® PROZYME ACTIVE, the disinfectant cleaner deconex® INSTRUMENT PLUS and the innovative automated 2-component system deconex® TWIN xpert. None of these products contain phosphates any more. Even our product for the basic cleaning and passivation of surgical instruments, deconex® 34 GR, is formulated to be phosphate-free. We are therefore making an important contribution to the preservation of our environment.
Literature
1) Sutton, M.A.; Bleeker, A.; Howard, C.M. et al. (2013): Our Nutrient World: The challenge to produce more food and energy with less pollution. Global Overview of Nutrient Management. Centre for Ecology and Hydrology, Edinburgh on behalf of the Global Partnership on Nutrient Management and the International Nitrogen Initiative.
2) Díaz, R.J. und Rosenberg, R. (2008): Spreading dead zones and consequences for marine ecosystems. Science 321:926-928
When are you going to set an ecological example and switch to phosphate-free products in the reprocessing of medical devices?


